This guidance will develop rapidly; please see the BHIVA website for the latest version
Monday 11 January 2021
COVID-19 vaccines
The UK was the first country to approve the use of a COVID-19 vaccine, which is being referred to as ‘The Pfizer vaccine,’ and vaccination with this started on the 8th December 2020. There is also another vaccine, known as ‘the AstraZeneca vaccine’ or ‘the Oxford vaccine,’ which has also been shown to be very effective at preventing COVID-19 and vaccination with this started on 4th January 2021. Other vaccines are also in the pipeline, including the Moderna vaccine, which was approved for use in the UK on 8th January 2021, with first vaccinations expected in the Spring.
The patient information leaflets for the three currently approved COVID-19 vaccines (Pfizer/BioNTech, AstraZeneca/Oxford and Moderna) all list a weakened immune system under the warnings and precautions for use, with Pfizer using HIV as an example. The leaflets advise affected patients to tell their doctor, nurse or pharmacist before vaccination. BHIVA has contacted Pfizer to challenge the mention of HIV in the leaflet.
This advice in the patient information leaflets is not based on concerns about safety. There is currently no evidence for more side effects in people with HIV. The guidance is there mainly because there is not yet very much research information about people with immune deficiency and/or HIV.
With some vaccines, people with HIV can produce a weaker response. We do not yet know if this is the case for the COVID-19 vaccines but they are expected to be protective in people with HIV and are recommended. The Department of Health recommends COVID-19 vaccination for all people with HIV, regardless of their CD4 count. None of the COVID-19 vaccines contain any live virus and so cannot cause COVID-19. The AstraZeneca/Oxford vaccine contains a cold virus, called adenovirus, which has been changed to make it inactive; similar adenovirus vaccines have been used and studied in populations where HIV is common and have been shown to be safe. All currently approved vaccines are thought to be safe for people with suppressed immune systems.
Is there any interplay between COVID-19 vaccine and HIV drugs?
No. HIV drugs do not affect the effectiveness of COVID-19 vaccines and COVID-19 vaccines do not affect how well HIV drugs work either. If you have had side effects or an allergic reaction to particular HIV drugs in the past, that does not mean you will get side effects from the COVID-19 vaccines.
There is no evidence that HIV drugs, taken as HIV treatment or prevention (PrEP) can be used to prevent or treat COVID-19. Some studies are looking into using HIV drugs to prevent COVID-19 but there are no results yet and no good evidence to suggest they will be effective.
Who will get the vaccine?
People will receive the vaccine in strict order of priority based on their age, health, occupation, whether they live in a care or residential home and who they live with. Vaccines will be offered strictly based on these priorities. There is no way to jump the queue, and you will be contacted when your vaccine is due.
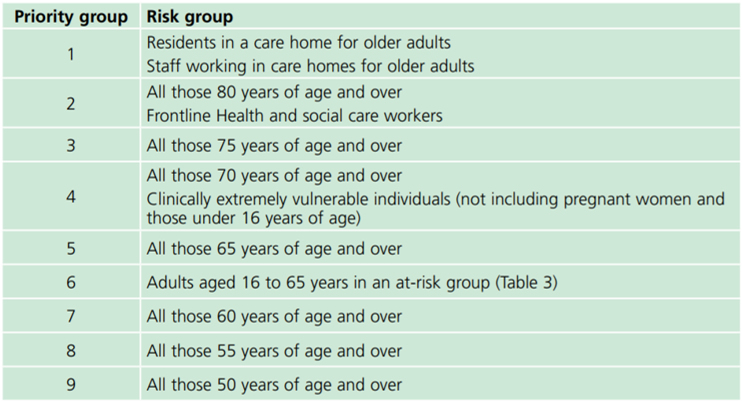
There are 9 priority groups: those in priority group 1 will get the vaccine first, followed by each in turn up until priority 9. After that the vaccine will be offered to everyone else (that is all the people not in priority groups 1-9).
Everyone with HIV is automatically in priority group 6 so you will get the vaccine earlier than many people. If your clinic thinks you are at higher risk you can be put into priority group 4, and get the vaccine sooner, but you will need to be added to a central NHS list to be put into priority group 4 - please contact your HIV clinic if you think this applies to you. We advise that the people at higher risk may include:
People with a CD4 count less than 50
People with a serious HIV-related illness (eg an opportunistic infection) in the last 6 months
People with a CD4 count between 50 and 200 with other issues that increase the risk of getting very sick, such as:
- Detectable viral load
- Low nadir CD4 (the lowest CD4 before starting HIV treatment)
- Other medical conditions associated with increased risk of severe COVID (such as asthma, COPD, diabetes, heart disease, kidney disease, liver disease, Parkinson’s disease, multiple sclerosis, motor neurone disease, conditions or drugs that suppress the immune system (e.g. steroid treatment), severe obesity.People with ‘multi-morbidity’ meaning that they have other health conditions that may increase the risk of getting very sick.
A full list of the different priority groups:
Will I have to have the vaccine?
No, nobody has to have the vaccine. However, we strongly recommend that everyone who is offered the vaccine accepts it. Although vaccine development has been very fast, it has been to the same high standards as any other vaccine trial and includes very careful monitoring of side effects. If anything changes we will update this guidance.
Where can I get the vaccine?
Currently vaccines are being offered in some hospitals, some pharmacies and in local vaccination centres run by GPs. Vaccines are not available in your local HIV clinic. You need to be registered with a GP in order to get a vaccination. Most people living with HIV are in priority group 6 which, for many people, means they are eligible for vaccination before other people of their age. However, if your GP does not know that you are HIV positive, you will not be offered the vaccination early. We have asked the national vaccine team to clarify how people who are not registered with a GP will be able to get the vaccine. We will update you on this and on any new plans to make the vaccine made more widely available from different places.
Does having the vaccine mean I don’t have to follow social distancing rules?
No. It will take many months to vaccinate everyone and it remains important to continue to wash your hands regularly, to wear a mask and to follow your local guidance about social distancing.
Notes:
1. More information is available on iBase https://i-base.info/qa/16330 which is updated regularly.
2. Safety data will be produced rapidly as the vaccine is rolled out. Please see MHRA website for the latest information.
For further information, please contact [email protected] or for media enquiries, please contact Jo Josh at [email protected] or +44 (0)7306 391875.



